NGSX Turnaround in full play!
|
Seite 1 von 3
neuester Beitrag: 18.04.13 17:41
|
||||
| eröffnet am: | 30.01.12 20:36 von: | thekey | Anzahl Beiträge: | 57 |
| neuester Beitrag: | 18.04.13 17:41 von: | Joschi307 | Leser gesamt: | 6259 |
| davon Heute: | 1 | |||
| bewertet mit 5 Sternen |
||||
|
Seite:

1
|
||||
|
--button_text--
interessant
|
|
witzig
|
|
gut analysiert
|
|
informativ
|
5
30.01.12 20:36
#1 NGSX Turnaround in full play!
Preis: $1.04
Website: http://www.neurogesx.com
Shares Out (Mil) 29.69
Float (Mil) 11.40
Großer Insiderkauf für $2.8M bei $1.65
Finviz:http://finviz.com/quote.ashx?t=ngsx
keydata: http://www.dailyfinance.com/quote/nasdaq/...x-inc/ngsx/key-statistics
Insider: http://www.insidercow.com/history/....jsp?company=NGSX&B1=Search!
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 20:38
#2 FDA Pending Catalysts For 5 Biotechs
NeurogesX, Inc. (NGSX)
NeurogesX is a biopharmaceutical company, focused on developing and commercializing pain management therapies. The company's lead product is Qutenza (capsaicin) 8% patch, which contains an 8% concentration of synthetic capsaicin.
The company, on February 9, should expect to hear from a FDA panel for review of Qutenza for additional indication of HIV Associated Peripheral Neuropathy.
I'll have to sit this one out until the stock starts perculating or further positive news is dispersed by management or independent trials.
http://seekingalpha.com/article/...alysts-for-5-biotechs?source=yahoo
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 20:47
#3 Finviz:
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 20:49
#4 insider buys and sells
0
30.01.12 20:50
#5 Analysten
0
30.01.12 20:52
#6 Robert Nelson
Dies wird sich allerdings in diesem Monat ändern imo
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 21:12
#7 kann bis 09.02 nach mehr drive aufnehmen
-----------
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
Optionen
| Antwort einfügen |
| Boardmail an "harry74nrw" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 21:41
#8 ...
Die Geschichte mit dem HIV vertraue ich nicht so.
Optionen
| Antwort einfügen |
| Boardmail an "Ggordie" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 21:51
#9 mhhh
1,50 - 2,00 wenn die Amis mehr schreiben und das volumen hoch bleibt
-----------
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
Optionen
| Antwort einfügen |
| Boardmail an "harry74nrw" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 22:14
#10 sk 1,08 + 19 %
nicht vergessen
sehr volatil
-----------
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
Optionen
| Antwort einfügen |
| Boardmail an "harry74nrw" |
|
Wertpapier:
NeurogesX Inc |
0
30.01.12 22:41
#11 Das Volumen
Das Volumen!
1.328.516 gehandelte Aktien heute!
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
1
30.01.12 22:43
#12 support / resistance
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
Angehängte Grafik:
screenhunter_03_jan.gif
![screenhunter_03_jan.gif]()
screenhunter_03_jan.gif
0
30.01.12 23:51
#13 ;-o
-----------
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
Optionen
| Antwort einfügen |
| Boardmail an "harry74nrw" |
|
Wertpapier:
NeurogesX Inc |
0
31.01.12 02:14
#14 Abwärtstrend
Also enormes Signal.
Das heutige volumen war auch ein Allzeitrekord bei einem steigenenden Kurs.
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
31.01.12 02:23
#15 February 9, 2012 und March 7, 2012
Qutenza is currently approved in the United States for the management of neuropathic pain associated with postherpetic neuralgia (PHN).
NeurogesX announced the FDA acceptance of the sNDA filing on November 14, 2011, and noted the Agency's goal is to review and act on the sNDA by March 7, 2012 under the Prescription Drug User Fee Act IIII (PDUFA). The sNDA seeks approval for a 30-minute application of Qutenza for the treatment of neuropathic pain associated with HIV-PN. The FDA granted the sNDA a priority six month review classification. FDA Priority Review status is given to drug candidates that offer significant advances in treatment, or provide a treatment where no adequate therapy exists, and accelerates the standard review time from 10 months to six months.
The Anesthetic and Analgesic Drug Products Advisory Committee (AADPAC) is an independent panel of experts that reviews and evaluates available data concerning the safety and effectiveness of marketed and investigational human drug products for use in anesthesiology and surgery, and makes appropriate recommendations to the Commissioner of Food and Drugs. While the FDA will consider recommendations of the AADPAC, the final decision regarding the approval of a product rests solely with the FDA.
The FDA will publish materials, including webcast information, pertaining to the meeting at http://www.fda.gov/AdvisoryCommittees/WhatsNew/default.htm.
Changes to the Advisory Committee meetings calendars may also be found on the FDA website at http://www.fda.gov/AdvisoryCommittees/Calendar/default.htm
About NeurogesX, Inc.
NeurogesX, Inc. (Nasdaq:NGSX - News) is a San Francisco Bay Area-based biopharmaceutical company focused on developing and commercializing novel pain management therapies. NeurogesX was founded on the concept that use of prescription-strength capsaicin could help manage the pain associated with neuropathic pain conditions. Since its inception, NeurogesX has leveraged its passion to help people with pain to efficiently develop this concept, resulting in the commercial launch of Qutenza (capsaicin) 8% patch in 2010. The Company continues to apply its knowledge and expertise in the development of other novel treatments for pain.
The Company's lead product, Qutenza, is a localized dermal delivery system containing prescription strength capsaicin that is currently approved in the United States and the European Union. Qutenza is now available in the United States for the management of neuropathic pain associated with postherpetic neuralgia (PHN). In Europe, Qutenza is being marketed by Astellas Pharma Europe Ltd. (Astellas), the European affiliate of Tokyo-based Astellas Pharma Inc., for the treatment of peripheral neuropathic pain in non-diabetic adults, either alone or in combination with other medicinal products for pain.
The Company has submitted a supplemental new drug application (sNDA) to expand the U.S. label for Qutenza for the management of neuropathic pain associated with HIV-associated peripheral neuropathy (HIV-PN) also referred to as HIV-associated neuropathy (HIV-AN) and HIV-distal sensory polyneuropathy (HIV-DSP).
The Company's most advanced product candidate, NGX-1998, is a topically applied liquid formulation containing a high concentration of capsaicin designed to treat pain associated with neuropathic pain conditions such as PHN. NGX-1998 has completed three Phase 1 clinical trials and one Phase 2 clinical trial in PHN patients.
The Company's early-stage pipeline includes pre-clinical compounds which include a number of prodrugs of acetaminophen. The Company has evaluated certain of these compounds in vitro and in vivo.
Safe Harbor Statement
This press release contains forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995 (the Act). NeurogesX disclaims any intent or obligation to update these forward-looking statements, and claims the protection of the Safe Harbor for forward-looking statements contained in the Act. Examples of such statements include but are not limited to statements regarding: the timing of the AADPAC meeting regarding the sNDA submission seeking expansion of the U.S. label for Qutenza to include management of neuropathic pain associated with HIV-associated peripheral neuropathy (HIV-PN); the timing of any FDA review of such sNDA, notwithstanding the March 7, 2012 PDUFA date; and the potential benefits of Qutenza. Such statements are based on management's current expectations, but actual results may differ materially due to various risks and uncertainties, including, but not limited to: difficulties or delays in the further development of Qutenza for additional indications, including difficulties or delays in receipt of FDA approval of the sNDA to expand the U.S. label for Qutenza for the management of pain due to HIV-PN; market acceptance of Qutenza in already approved indications may not be sufficient to support further pursuit of an expanded label for Qutenza, including as a result of physician or patient reluctance to use Qutenza; Qutenza and NeurogesX' other product candidates may have unexpected adverse side effects; and unexpected or increased expenses in the commercialization and continued development of Qutenza or the development of NGX-1998. For further information regarding these and other risks related to NeurogesX' business, investors should consult NeurogesX' filings with the Securities and Exchange Commission.
Contact:
NeurogesX, Inc.
Stephen Ghiglieri
Executive Vice President, COO
and CFO
(650) 358-3310
sghiglieri@neurogesx.com
The Ruth Group
Stephanie Carrington (investors)
(646) 536-7017
scarrington@theruthgroup.com
Victoria Aguiar (media)
(646) 536-7013
vaguiar@theruthgroup.com
http://finance.yahoo.com/news/...-Confirms-FDA-pz-2414556104.html?x=0
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
1
31.01.12 02:26
#16 Pipeline der Produkte
http://www.neurogesx.com/pipeline

Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
31.01.12 02:29
#17 Pipeline der Produkte
Total Cash (mrq): $45.23M
Total Cash Per Share (mrq): $1.52
Total Debt (mrq): $65.13M
Total Debt/Equity (mrq): N/A
Current Ratio (mrq): 2.60
Book Value Per Share (mrq): $-1.97
http://finance.yahoo.com/q/ks?s=NGSX+Key+Statistics
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
31.01.12 09:01
#18 USA ab 15:30
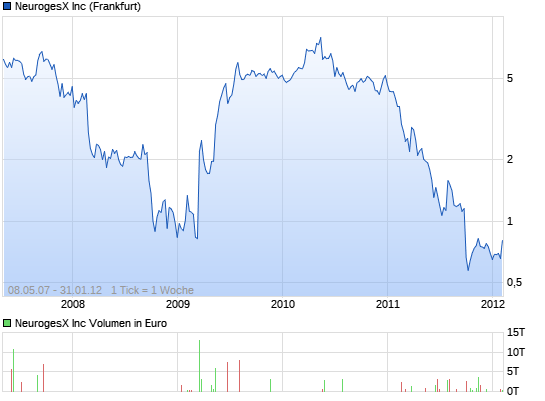
da kann noch was gehen
-----------
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
Optionen
| Antwort einfügen |
| Boardmail an "harry74nrw" |
|
Wertpapier:
NeurogesX Inc |
0
02.02.12 01:24
#19 Announces $3.0 Million Private Placement
SAN MATEO, Calif., Feb. 1, 2012 (GLOBE NEWSWIRE) -- NeurogesX, Inc. (Nasdaq:NGSX - News), a biopharmaceutical company focused on developing and commercializing novel pain management therapies, today announced that on January 31, 2012 it entered into a securities purchase agreement in connection with a private placement to a group of institutional accredited investors and, subject to standard closing conditions, is expected to receive approximately $3.0 million in gross proceeds from the sale of 2,969,685 common shares at $1.01 per share based on the consolidated closing bid price of NGSX common stock on the NASDAQ Global Market on January 31, 2012. The funding was led by a large, global institutional investor and included certain other existing investors. The funding was initiated by an inbound inquiry.
The securities sold in the private placement have not been registered under the Securities Act of 1933, as amended, or state securities laws and may not be offered or sold in the United States absent registration with the Securities and Exchange Commission or an applicable exemption from the registration requirements. NeurogesX has agreed to file a registration statement with the Commission covering the resale of the shares of common stock sold in the private placement.
This press release shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction.
About NeurogesX, Inc.
NeurogesX, Inc. (Nasdaq:NGSX - News) is a San Francisco Bay Area-based biopharmaceutical company focused on developing and commercializing novel pain management therapies. NeurogesX was founded on the concept that use of prescription-strength capsaicin could help manage the pain associated with neuropathic pain conditions. Since its inception, NeurogesX has leveraged its passion to help people with pain to efficiently develop this concept, resulting in the commercial launch of Qutenza (capsaicin) 8% patch in 2010. The Company continues to apply its knowledge and expertise in the development of other novel treatments for pain.
The Company's lead product, Qutenza, is a localized dermal delivery system containing prescription strength capsaicin that is currently approved in the United States and the European Union. Qutenza is now available in the United States for the management of neuropathic pain associated with postherpetic neuralgia (PHN). In Europe, Qutenza is being marketed by Astellas Pharma Europe Ltd. (Astellas), the European affiliate of Tokyo-based Astellas Pharma Inc., for the treatment of peripheral neuropathic pain in non-diabetic adults, either alone or in combination with other medicinal products for pain.
The Company has submitted a supplemental new drug application (sNDA) to expand the U.S. label for Qutenza for the management of pain due to HIV-associated peripheral neuropathy (HIV-PN), previously referred to as HIV-associated neuropathy (HIV-AN) and HIV-distal sensory polyneuropathy (HIV-DSP).
The Company's most advanced product candidate, NGX-1998, is a topically applied liquid formulation containing a high concentration of capsaicin designed to treat pain associated with neuropathic pain conditions such as PHN. NGX-1998 has completed three Phase 1 clinical trials and one Phase 2 clinical trial in PHN patients.
The Company's early-stage pipeline includes pre-clinical compounds which include a number of prodrugs of acetaminophen. The Company has evaluated certain of these compounds in vitro and in vivo.
Safe Harbor Statement
This press release contains forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995 (the Act). NeurogesX disclaims any intent or obligation to update these forward-looking statements, and claims the protection of the Safe Harbor for forward-looking statements contained in the Act. Examples of such statements include but are not limited to statements regarding: the potential expansion of the U.S. label for Qutenza to include management of pain due to HIV-associated peripheral neuropathy (HIV-PN); the expected closing, and proceeds from, the private placement transaction and the registration of shares issued in connection with the private placement with the U.S. Securities and Exchange Commission. Such statements are based on management's current expectations, but actual results may differ materially due to various risks and uncertainties, including, but not limited to: difficulties or delays in the further development of Qutenza for additional indications, including difficulties or delays in receipt of FDA approval of the sNDA to expand the U.S. label for Qutenza for the management of pain due to HIV-PN; market acceptance of Qutenza in already approved indications may not be sufficient to support further pursuit of an expanded label for Qutenza; Qutenza, NGX-1998 and NeurogesX' other product candidates may have unexpected adverse side effects; and difficulties or delays in the closing of the private placement and subsequent registration of shares issued in connection with such private placement. For further information regarding these and other risks related to NeurogesX' business, investors should consult NeurogesX' filings with the Securities and Exchange Commission.
Contact:
NeurogesX, Inc.
Stephen Ghiglieri
Executive Vice President, COO and CFO
(650) 358-3310
sghiglieri@neurogesx.com
The Ruth Group
Stephanie Carrington (investors)
(646) 536-7017
scarrington@theruthgroup.com
Victoria Aguiar (media)
(646) 536-7013
vaguiar@theruthgroup.com
http://finance.yahoo.com/news/...Announces-3-0-pz-1361370383.html?x=0
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
02.02.12 22:15
#20 5 % rauf
-----------
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
.ariva.de/forum/Lexam-VG-Gold-WKN-A1H396-Faktensammlung-430036
Optionen
| Antwort einfügen |
| Boardmail an "harry74nrw" |
|
Wertpapier:
NeurogesX Inc |
0
04.02.12 14:33
#21 Interessanter Wert! Immer für eine Verdopplung
Optionen
| Antwort einfügen |
| Boardmail an "martin30sm" |
|
Wertpapier:
NeurogesX Inc |
0
06.02.12 13:17
#22 FDA Entscheidung am 09.02.
Optionen
| Antwort einfügen |
| Boardmail an "Joschi307" |
|
Wertpapier:
NeurogesX Inc |
0
06.02.12 17:02
#23 Nächste Kursziel sollte im Bereich von $1.50
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
06.02.12 18:50
#24 IHUB Membermark 33
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
0
06.02.12 19:04
#25 Tageshoch $1.20
Optionen
| Antwort einfügen |
| Boardmail an "thekey" |
|
Wertpapier:
NeurogesX Inc |
Seite:


1









